less than the first ionization potential of Mg (b) The second ionization potential. periodically when elements are arranged according to increasing atomic number. The greater the distance, the less the attraction and so the less energy is released as electron affinity. Arrange the elements with the following electronic configurations in. the elements is a product of the first periodic table published bydmitri. 
Largest radius Smallest radius Answer Bank Na 02- Mg2 F N3- Arrange the atom and ions according to radius. 
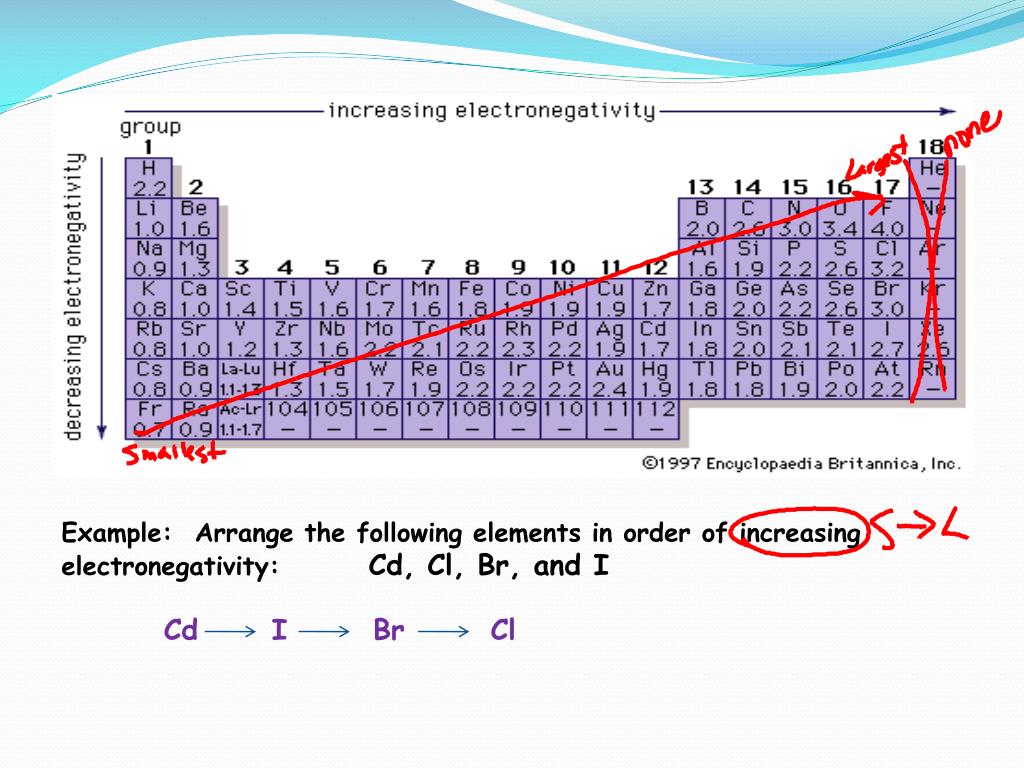
Therefore, given elements are arranged from highest to least ionization energy as follows. Rb being the largest in size will have low ionization energy whereas Li being the smallest will have highest ionization energy. Highest ionization energy Lowest ionization energy Answer Bank Ge Si C Sn Arrange these ions according to ionic radius. For example, Na, Li, Rb and K are all group 1 elements. The over-riding factor is therefore the increased distance that the incoming electron finds itself from the nucleus as you go down the group. Question: Arrange the elements according to first ionization energy. This will be approximately the same in both these cases and so does not affect the argument in any way (apart from complicating it!). arrowforward Write equations corresponding to the following: a. There is also a small amount of screening by the 2s electrons in fluorine and by the 3s electrons in chlorine. (c) Arrange the three elements B, Al, and C in order of increasing first ionization energy. In recent years, global climate change has been abnormal, resources are increasingly depleted, and countries are accelerating the development of new energy sources 1,2,3. Successive ionization energies, in attojoules per atom, for the new element are given in the table. 1) Arrange the following elements in order of increasing atomic size: Al, B. Energy is the cornerstone of economic and social development and a fundamental guarantee for the survival of human society. But again the incoming electron feels a net attraction from the nucleus of 7 (17 protons less the 10 screening electrons in the first and second levels). Arrange these elements according to first ionization energy (highest to lowest) Ne F O N C Li Be B Ne, F, N, C, Be, B, Li A space probe identifies a new element in a sample collected from an asteroid. Well, since ionization energy increases across a period, Sn will have a smaller ionization energy than Te. 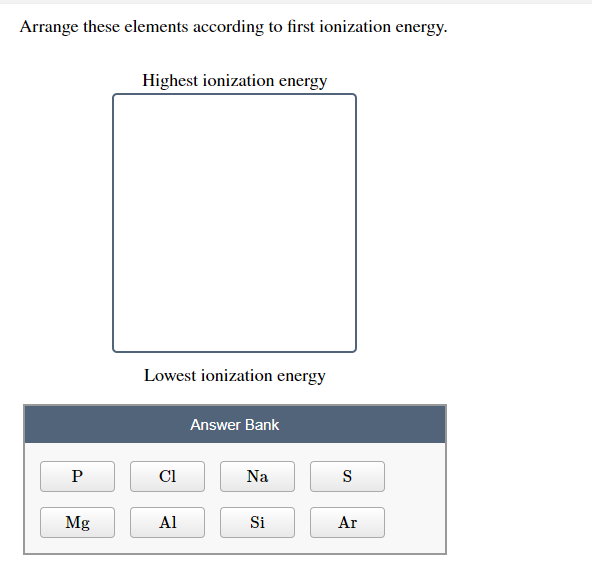
The period above (5) has two of the elements: Sn and Te. radius ( III ) B < C < N < O Increasing first ionization energy 44. In contrast, chlorine has the electronic structure 1s 22s 22p 63s 23p x 23p y 23p z 1 with 17 protons in the nucleus. P b is the element that is in the lowest period at 6 (and lowest group at 14) in the periodic table it's the smallest ionization energy. In which of the following arrangements, the order is according to the property. It therefore feels a net attraction from the nucleus of 7 (9 protons less the 2 screening electrons). It has 9 protons in the nucleus.The incoming electron enters the 2-level, and is screened from the nucleus by the two 1s 2 electrons. The correct order of first ionization energy should be: K < Na < Li. Each successive IE is larger than the previous because an electron is being removed from an atom with a progressively larger positive charge. ChlorineĪ fluorine atom has an electronic structure of 1s 22s 22px 22py 22pz 1. According to Wikipedia Electron affinity, In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as the amount of energy released when an electron is added to a neutral atom or molecule in the gaseous state to form a negative ion. First Ionization Energy (IE 1 ): A ( g) A ( g) e Second Ionization Energy (IE 2 ): A ( g) A 2 ( g) e Third Ionization Energy (IE 3 ): A 2 ( g) A 3 ( g) e and so forth. (g) e^- \nonumber \]Įach successive IE is larger than the previous because an electron is being removed from an atom with a progressively larger positive charge.\): Fluorine vs.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
